In the world of advanced medical devices, the synergy between a device and a therapeutic drug can elevate patient care to new heights. Drug-coated devices, from cardiovascular stents to implantable systems, promise targeted treatment with controlled-release properties. However, achieving this is a complex, high-stakes process. It demands a fusion of precision engineering, sophisticated formulation, and rigorous regulatory adherence to ensure the device is not only effective but also safe and reliable over its entire lifecycle.
For medical device innovators, mastering the coating process is non-negotiable. Whether you’re in early-stage R&D or scaling up for commercial launch, focusing on the following seven critical areas will pave the way for success.
1. The Art of Uniformity: Precision in Every Layer

The effectiveness of your device hinges directly on the precision of its coating. An uneven coat can lead to unpredictable drug dosages and compromised therapeutic outcomes.
The solution lies in meticulous application technology, such as micro-volume dispensing or advanced spray coating systems, which maintain exacting control over flow rates and thickness.
For complex geometries like stents or implants, ensuring complete and uniform coverage through strategic rotation or movement during application is essential.
2. Perfecting the Payout: Controlled Drug Release
A controlled-release coating must deliver the drug precisely as intended over a specific duration.
Any deviation—be it overdosing, underdosing, or premature depletion—can undermine the device’s therapeutic purpose.
Success requires a deep understanding of formulation science, from optimizing polymer matrices like PLGA to fine-tuning drug-to-polymer ratios and curing conditions.
Advanced techniques, such as using multilayer coatings or diffusion barriers, can also be employed to achieve the desired elution profile.
3. Built to Last: Superior Coating Adhesion
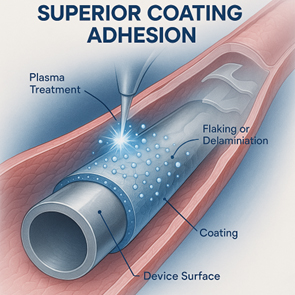
Inside the dynamic environment of the human body, your coating must stay put.
Poor adhesion can lead to delamination or flaking, which not only causes premature drug release but can also pose significant safety risks.
Achieving rock-solid adhesion starts with meticulous surface preparation of the device, including advanced cleaning or plasma treatment.
It’s also vital to select coating materials that are inherently compatible with the device’s substrate and to use adhesion-promoting agents where necessary.
4. Safety First: Biocompatibility and Stability
Patient safety is paramount.
Every component of the coating and the drug it releases must be biocompatible and stable from the moment of manufacture to the completion of its therapeutic mission.
This requires using well-characterized, regulatory-approved biomaterials and conducting exhaustive studies on chemical stability and the impact of sterilization.
A critical part of this process is ensuring that no harmful leachables or degradation byproducts are generated that could compromise patient safety.

5. From Lab to Life: Repeatable and Scalable Processes
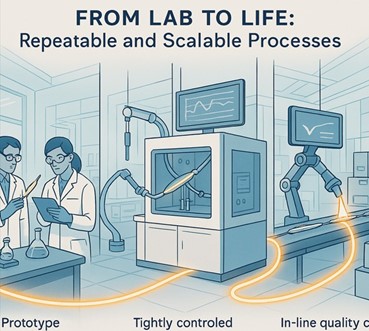
A brilliant prototype is only valuable if it can be manufactured consistently at scale.
Your manufacturing process must be robust, repeatable, and ready for commercial production without any drop in quality.
This is achieved by using validated, automated coating systems with tightly controlled parameters and integrating in-line quality control checks for critical attributes like coating weight and uniformity.
6. Navigating the Maze: Regulatory Compliance
Drug-coated devices are classified as combination products, placing them under the stringent oversight of both medical device and pharmaceutical regulations (e.g., FDA, EMA).
Navigating this complex landscape requires a proactive approach.
Adherence to GMP, ISO 13485, and ICH standards is not optional.
You must be prepared for intense scrutiny with comprehensive documentation covering everything from process validation to clinical safety and efficacy data.

7. The Final Proof: Rigorous Functional Testing

Ultimately, you must prove that your coated device works as intended in real-world conditions.
This final validation step involves a battery of tests.
In vitro release studies are conducted to confirm the drug elution profile, while mechanical tests ensure the coating’s integrity.
Crucially, testing protocols should simulate physiological environments, by using flow loops or controlling for body temperature and pH, to generate data that truly reflects in-vivo performance.
Conclusion: The Promise of Precision
Developing a successful drug-coated medical device is a journey of precision, diligence, and expertise.
By focusing on these seven essential factors, you can navigate the technical and regulatory challenges to create a product that is safe, effective, and reliable.
The goal is clear: to deliver the right dose to the right place at the right time, transforming patient outcomes and setting a new standard in medical innovation.
